Intra-phage immunity: Set a (pro)phage to catch a phage
E. coli prophages encode an arsenal of defense systems to protect against temperate phages
The classic view of phage biology has long cast the battle as phage versus bacterium. But the real picture is more complex. Mobile genetic elements (MGEs), such as phages, plasmids, and transposons, do not just attack bacteria; they can engage in direct conflict with one another using bacteria as the playground. In this issue of Cell Host & Microbe, Brenes and Laub dive into this less-explored layer of conflict, carrying out one of the first large-scale, systematic efforts to uncover defense systems encoded specifically by temperate phages.
There has been major progress in cataloguing bacterial defenses, from classical CRISPR-Cas and restriction-modification systems to a growing repertoire of other systems in so-called defense islands. These efforts, however, have largely focused on protection against lytic phages. By contrast, defense systems that target temperate phages—or are carried by them—have received far less attention. A few studies have hinted that prophages might defend their hosts against other temperate phages, but a systematic search across a broad collection of temperate phages had not previously been undertaken.
Temperate phages present a unique evolutionary challenge. Once integrated into a bacterial genome, their survival becomes linked to that of the host. It is therefore logical that prophages would want to keep out competitors. Classic superinfection exclusion systems do just that, blocking closely related phages from entering or replicating. But prophage-encoded systems that provide broader immunity against other, unrelated temperate phages have been harder to detect and characterize.
To address this gap, Brenes and Laub focused explicitly on temperate phages as both potential sources and targets of anti-phage defenses. They induced 33 temperate phages from a diverse panel of E. coli strains, generated single-lysogen libraries in three host backgrounds, and challenged each with the remaining phages. The result was a dense web of phage-phage inhibition, with over 75% of prophages providing protection against at least one other phage, and often many. Through an iterative deletion approach, the authors mapped 17 distinct defense systems responsible for these interactions. Of these, only four had been earlier described, like HK002 Nun and HK97 Gp15. The other 13 were previously unreported, five of which turned out to be bona fide toxin-antitoxin (TA) modules.
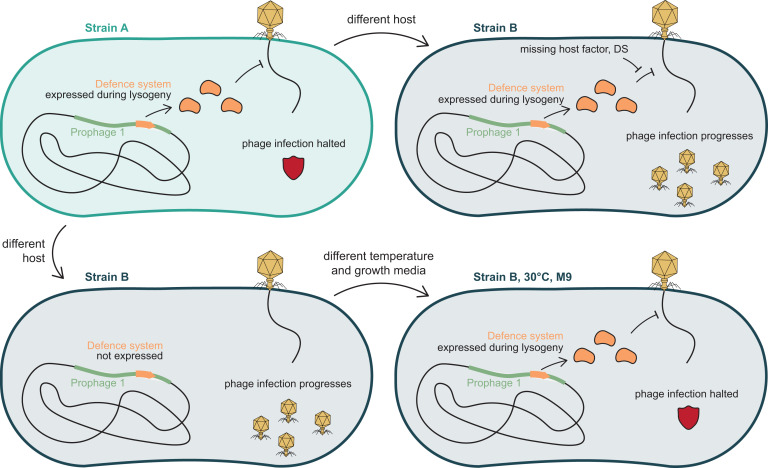
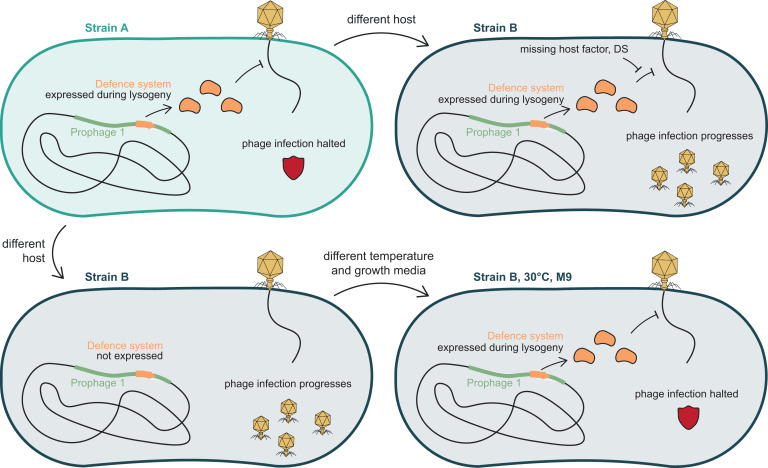
These results reinforce the idea that temperate phages are not just passengers; rather, they can actively shape which other phages are allowed in. Several of the systems discovered here were broad-acting, while others had narrow, phage-specific effects. Some were validated as superinfection exclusion systems, including one resembling the T4 spackle protein. Others were more complex, including TA systems such as HipA-HipD and a tripartite HicABC system that likely senses phage infection via a chaperone component. Interestingly, some of these systems were located in late lytic operons but were detectably expressed during lysogeny, suggesting leaky expression or dedicated regulatory elements that allow them to function even when most prophage genes are dormant.
Interestingly, the authors found that defense strength varied not only with the system itself but also with genetic and environmental context. Defense phenotypes were often stronger in the E. coli ECOR13 background than in E. coli K-12, and many were more active in minimal medium at a lower temperature. In some cases, these differences were not explained by transcriptional changes, pointing instead to post-transcriptional regulation or interactions with host factors or other defenses. This has implications for how prophage-encoded defense may behave when horizontally transferred, since systems that are active in one background might be inert in another, and vice versa.
The work raises several follow-up questions. Are these systems exclusive to temperate phages, or can they also block lytic ones? Since many of the tested phages were in their lytic cycle, there is a good chance that they are also active against obligately lytic phages. Could some prophage-encoded systems specifically target the lysogenic cycle, for example, by preventing integration, recombination, or excision? Most work on bacterial immunity focuses on lytic infection, but the lysogenic cycle could offer a different set of defense opportunities.
Taken together, this work provides a model for targeted defense discovery in phage genomes. It reframes temperate phages not just as latent threats or genetic hitchhikers but also fierce competitors in their own right. By systematically mapping defense systems hidden in prophages, Brenes and Laub expand our understanding of immunity in microbial communities. As more attention turns to the defense potential of MGEs, it may—proverbially—take a phage to catch a phage.
Read here: https://doi.org/10.1016/j.chom.2025.05.012