BiomX Inc.: Clinical Stage Phage Therapeutics Company Looks Promising
Summary
- BiomX is a clinical-stage microbiome company using its novel BOLT phage therapy platform to develop customized phage therapies to treat chronic diseases ranging from IBC, colorectal cancer, Cystic Fibrosis and acne.
- 6 programs in its pipeline. Positive Ph 1 data for BX001 in acne. Positive Ph 1a pharmacokinetic data for IBD/PSC evaluating oral delivery. Clinical trial read outs in 2021 and 2022.
- Collaboration agreements with Janssen, Boehringer Ingelheim and a leading cosmetics company. Investors include Orbimed, Takeda, and J&J.
- Market cap of approximately $170 million with approximately $50 million cash. While BiomX is in early stage, its phage therapy technology looks very promising and if proven out, BiomX could generate substantial upside for investors.
- In December, BiomX has filed a $150 million base prospectus with the SEC as well as a $50 million “at the market” prospectus dated December 4, 2020 with Jefferies LLC, as agent, so an equity financing in 2021 is certainly a possibility.

On January 28 and 29, I (virtually) attended the 5th Microbiome Movement- Drug Development Summit Europe 2021 conference (“2021 Microbiome Europe Conference”). I listened to two days of microbiome therapeutic company-related presentations and talks on microbiome therapeutics.
The last live webcast presentation was given by an Israel-based clinical-stage biotech company, BiomX Inc. (NYSE, TASE: PHGE) developing natural and engineered phages to target pathogenic bacteria.
I subsequently listened to a webcast presentation made by BiomX’s CEO Jonathan Solomon on January 11, 2021 at the virtual HC Wainwright Bioconnect virtual conference, currently available on BiomX’s website (approximately 27.5 minutes in length).
For additional background on BiomX, I also recommend reading the November 24, 2020 article published on Biospace.com, “From Acne to Cancer, BiomX Infects Bad Bacteria with Modified Viruses“.
Investment Thesis
BiomX Inc. (PHGE) is an Israeli microcap clinical-stage biotech company utilizing proprietary next-generation synthetic biology platforms and technologies, (including its high-resolution DNA analysis, AI, cloud computing and other computational tools) to develop phage cocktails (both naturally occurring and engineered) as a novel mode of action to treat various chronic and other diseases, including those that have become resistant to current antibiotics. Early pre-clinical and clinical results have been promising.
Shareholders include Orbimed, Takeda and Johnson & Johnson.
BiomX has also entered into collaboration agreements with Janssen, Boehringer Ingelheim, and others. BiomX’s market cap is approximately $170 million with nearly $50 million in cash or equivalents as of September 30, 2020).
In the author’s opinion, BiomX has enormous commercial value and potential and is currently undervalued.
Harnessing Phages as Natural Predators of Bacteria
As described on the BiomX website, Bacteriophages or Phages are viruses that form part of the microbiome which are “natural predators of bacteria” that “target and rapidly kill their bacterial hosts”. One of the key advantages of Phages is that they “are specific to particular bacterial species or strains, targeting and killing them without disrupting other neighboring, potentially beneficial, bacteria.”
Source: BiomX February 2, 2021 Corporate presentation
The effectiveness of phage cocktails as a therapeutic treatment received a lot of attention a few years ago when a number of patients with life-threatening infections were cured by phage cocktails.
A number of examples are cited in a March 20, 2020 Chardan Capital Markets Industry Note Chardan_Micrombiome_Biotechnology Pharmaceuticals_2020-03-20.pdf following Chardan’s 2nd Annual Microbiome Summit held last March 16, 2020, include:
- a professor who was in a coma after suffering a near-fatal multidrug-resistant strain of Acinetobacter baumannii, and improved “almost immediately” after treatment with an experimental phage cocktail”;
- a patient treated with a phage cocktail that cleared a years-long chronic infection that was successfully cleared, allowing a heart transplant surgery; and
- a 15-year-old cystic fibrosis patient who was successfully treated with a cocktail of 3 phages after contracting a super-resistant mycobacterium infection after receiving a double lung transplant.
In response to an email inquiry from me, last week BiomX CBO Assaf Oron sent me a 9 slide BiomX presentation on how phage therapies have been used during the past few years on patients on a compassionate use basis. I will refer to that 9 slide presentation as the “Compassionate Use presentation“.
In the slide below taken from the Compassionate Use presentation, BiomX summarizes the approximate 100-year timeline from when Phages were discovered in 1919 to examples of phage treatments (given on a compassionate use basis to patients from 2015 to 2019) to the 2020 positive Phase 1 data from BiomX phage acne product.
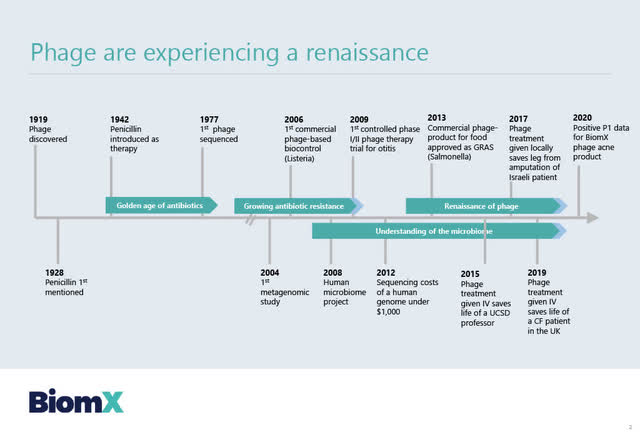
Source: BiomX: Compassionate Use presentation
Phage Safety Profile
Phages have also been shown to have a very safe profile. See the slide below for more details.
Source: BiomX Compassionate Use presentation
BiomX Use of Synthetic Biology to Develop Phage Cocktails
While there is no single definition of synthetic biology, according to the NIH National Human Genome Research Institute website,
Synthetic biology is a field of science that involves redesigning organisms for useful purposes by engineering them to have new abilities…”
BiomX is using proprietary ‘synthetic biology’ technology platforms to develop phage cocktails, composed of both natural and engineered phages, to target and destroy harmful bacteria in 6 disease target programs including:
1. acne;
2. inflammatory bowel disease (“IBD”);
3. primary sclerosing cholangitis (“PSC”);
4. cystic fibrosis (“CF”);
5. atopic dermatitis; and
6. colorectal cancer.
Licensing Agreement with MIT and Weizmann Institute’s Yeda Research and Development agency: According to BiomX’s Form 10-Q filed November 12, 2020, with the SEC (available on BiomX’s website), BiomX Israel obtained exclusive royalty-bearing patent licensing rights from the Massachusetts Institute of Technology to “synthetically engineer phage”. BiomX has also entered into one or more licensing agreements with the Weizmans’ Institute’s Yeda Research and Development agency.
Personalized GMP Grade Phage Cocktails: In his January 11th webcast, Mr. Solomon described how, if a sample of bacteria from a patient is provided, BiomX has the technology to create a GMP grade phage cocktail to treat that individual’s specific bacteria within 6 to 8 weeks.

Source: BiomX February 2, 2021 Corporate presentation
Phage Cocktail Therapies Can Be used to Target Specific Bacteria and both Large Market & Orphan Diseases
BiomX’s phage cocktails can be designed to target specific bacteria (which are known to cause certain diseases) and to avoid bacterial resistance. BiomX’s overall plan is to develop a pipeline of phage-based therapies to address both large-market and orphan diseases in multiple applications as set out in BiomX’s slide below.
XMarker Platform: “Utilizing High-Resolution DNA Analysis, AI and High-scale Cloud Computing to Discover Predictive Microbial Genomic Signatures”
As set out in its website describing its technology, BiomX has a “first-of-its-kind” XMarker platform which uses a “unique metagenomics-based approach to discover predictive microbial genomic signatures that can be further developed into biomarkers. The platform combines ultra-high-resolution DNA analysis, AI techniques and high-scale cloud computing resources to build classifiers of high sensitivity and specificity.”
BiomX has entered into collaboration agreements with both Janssen and Boehringer Ingelheim to use BiomX’s XMarker platform to discover biomarkers in IBD.
Source: BiomX February 2, 2021 corporate slide presentation
BOLT Platform: On November 12, 2020, BiomX unveiled its BOLT platform (“BacteriOphage Lead to Treatment”) “comprised of proprietary computational tools, automated screening, synthetic engineering capabilities and various validation assays aimed at developing natural or engineered phage cocktails that target a particular pathogenic bacteria.”
As summarized on BiomX’s slide below, the Bolt phage discovery platform allows the creation of both:
- a personalized phage treatment (targeted to the specific strain of a person) on an accelerated 6 to 8-week basis; and
- an optimized phage therapy by treatment indication (1 to 2 years).
Source: BiomX February 2, 2021 corporate presentation
BiomX Inc. Corporate Summary
Trading symbol: NYSE, TASE: (PHGE)
Head Office: Ness Ziona, Israel
Warrants trade under symbol: (PHGE.WS). Two warrants need to be exercised to acquire 1 common share in BiomX (PHGE). These warrants expire in October 2014 with a strike price at $11.50 and are subject to various conditions that an investor should read before purchasing.
A BiomX combined common share and single warrant trade under the symbol (PHGE.U)
BiomX Inc. BiomX, formerly Chardan Healthcare Acquisition Corp. (“CHAC”), was originally incorporated in Delaware on November 1, 2017. On July 16, 2019 CHAC entered into an agreement to merge with BiomX Israel. The company was renamed October 28, 2019 as BiomX Inc. See Form 10-Q filed with the SEC on November 12, 2020, for further details.
Institutional investors include Orbimed, Takeda and Johnson & Johnson
Partnerships/ Collaborations with Janssen and Boehringer Ingelheim. Collaboration for BiomX’s acne-focused therapeutic (BMX001) with a major (unidentified) global cosmetic company.
Shares outstanding: 23,170,000 common shares as of September 30, 2020 (as per Form 10-Q filed with the SEC).
Options outstanding: 3,671,549 options are outstanding with a weighted average strike price of $3.08 (as per Form 10-Q filed with the SEC).
Warrants Outstanding: Approximately 7 million warrants outstanding of which 6.4 million (each warrant can be exercised for 1/2 of a common share). The warrants have an exercise price of $11.50 subject to various conditions set out in Form 10-Q. 3,500,000 of these $11.50 warrants are considered public and are traded either under the symbol (PHGE.WS) or as part of a Unit with a common share under the symbol (PHGE.U).
BiomX Inc. 52 week range: (PHGE) share price in $US trading range: $4.52 to $11.05. Currently $7.43as of Tuesday February 17.
Market Cap: Approximately $170 million based upon outstanding common shares.
Cash on cash equivalents: Approximately $53.3 million as of September 30, 2020. See Form 10-Q dated as of September 30, 2020 filed November 12, 2020 with the SEC for more financial details.
Prospectus: On December 4, 2020, BiomX entered into a $50 million “at the market” ATM facility with Jefferies LLC. See Form 8-K for more details. BiomX has also filed a $150 million shelf prospectus in December 2020.
Management Team
BiomX has an experienced senior management team. The summary below is taken from BiomX website Executive Team summary:
CEO Jonathan Solomon, MBA (Harvard), M.Sc. electrical engineering (Tel Aviv University), undergraduate degree in Physics and Mathematics (Hebrew University in Jerusalem). Mr. Solomon was previously a “co-founder, president, and CEO of ProClara (formerly NeuroPhage)” a biotech company focused on treating neurodegenerative diseases. Previously, Mr. Solomon was a member of the Israeli Defense Forces ‘Talpiot’ program.
As an aside, members of Israel’s Talpiot program are an elite group within the Israeli Defense Forces who are credited with some of Israel’s most advanced military and scientific breakthroughs. Readers might find this article about Talpiot of interest.
Chief Business Officer Assaf Oron M.Sc. in Biology (bioinformatics), B.Sc. in Chemistry and Economics. Prior to joining BiomX in January 2017, he was Exec. VP of Corporate Development and Executive VP of strategy and business development at Evogene (EVGN) where he was employed for 10 years.
Chief Medical Officer Dr. Sailaja Puttagunta, M.D., formerly VP Development at Iterum Therapeutics which developed antibiotics against “multi-drug resistant pathogens.” Previously, she served as VP, Medical Affairs for Anti-infectives at Allergan. She is a graduate of the Gandhi Medical College in India and completed her residency in Internal Medicine and a fellowship in Infectious Diseases at Yale University School of Medicine.
Source: BiomX February 2, 2021 Corporate Presentation
Board of Directors
The backgrounds of BiomX’s board of directors are summarized below. Additional biographical details are available for each board member on the Company’s website.
Chairman: Russell G. Greig, Ph.D. formerly President of SR One, GlaxoSmithKline’s corporate venture group.
Jonathan Solomon: President & CEO of BiomX
Dr. Gbola Amusa, MD, Partner, Director of Research and Head of Healthcare Research at Chardan Capital.
Jonas Grossman, President of Chardan Capital.
Alan Moses, M.D., Co-founder and co-director of the Clinical Investigator Training Program at Beth Israel Deaconess-Harvard Medical School-MIT. Dr. Moses served as Senior Vice President and Chief Medical Officer of the Joslin Diabetes Center in Boston. He was appointed Professor of Medicine at Harvard Medical School. Previously Dr. Moses worked at the National Institutes of Health and completed subspecialty clinical training in Endocrinology.
Paul Sekhri, President and CEO of gene-editing company eGenesis. Previously, he was President and CEO of Lycera Corp., and served as Executive Vice President of Business Development and Chief Strategy Officer for Teva Pharmaceuticals.
While not mentioned in his biography on the BiomX website, Mr. Sekhri is also the Chairman of the Board of Directors of Compugen Ltd. (CGEN), another innovative Israeli biotech company that I have written about extensively.
Lynne Sullivan, formerly CFO of Compass Therapeutics, LLC. Prior to Compass, Ms. Sullivan was the Sr. VP of Finance for Biogen, Inc.
Source: BiomX February 2, 2021, Corporate presentation
BiomX Pipeline
BiomX’s two most advanced programs involve BX001 for the treatment of acne, discussed in more detail below (partnered with an unidentified major cosmetics company), and BX003 for the treatment of IBD and PSC.
Source: BiomX February 2, 2021 corporate presentation
1. BX001 – A Phage Cocktail Being Clinically Tested against C. acnes Strains
As explained by Solomon and described on BiomX website describing its pipeline, BX001 is a cocktail of naturally-occurring phages that targets Cutibacterium acnes (“C. acnes”). BX001 is in gel form that is used topically to improve the appearance of acne-prone skin.
As set out in BiomX slide below, BX001 appears to be “active against 96% of tested C. acnes clinical strains (in vitro)”, penetrates biofilm and is active against antibiotic-resistant strains.
The current standard of care by dermatologists in the treatment of acne is the extensive use of topical and oral antibiotics.
As described in BiomX website there has been a serious increase in antibiotic resistance due to the overuse of antibiotics during the past number of decades.

Source: BiomX February 2, 2021 corporate presentation
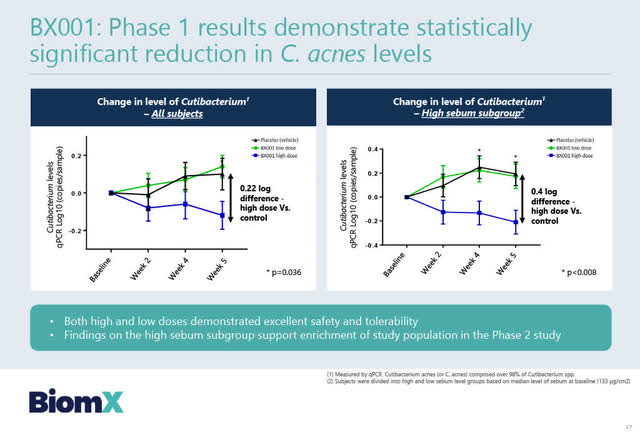
BX001 Positive Phase 1 Clinical Results
On March 31, 2020, BiomX announced positive top-line Phase 1 results from its cosmetic clinical study of BX001 which demonstrated “significant reductions in C. acnes levels” in the “75 enrolled patients with mild-to-moderate acne. … The study met its primary endpoint of safety and tolerability for both doses of BX001, as well as a statistically significant (p=0.036) reduction of C. acnes levels for the high dose of BX001 compared to placebo.”
The slide below sets out BX001 Phase 1 clinical results.
Source: BiomX February 2, 2021 Corporate presentation
BiomX’s Phase 2 cosmetic clinical study results of BX001 (12-week, double-blind, placebo-controlled, 100 patients with mild-to-moderate acne) are expected to be released in Q2 2021. (Source: BiomX February 2, 2021, corporate presentation)
BX001 does not require FDA or equivalent European approval for the treatment of acne in order to commercially launch as the studies are being conducted as cosmetic clinical. The BX001 Phase 2 study is being conducted in order to substantiate claims to support future marketing and sales, and to obtain “premium pricing”.
In response to my email questions regarding BX001, CBO Assaf Oron clarified in a February 14th email to me (which I have summarized below) that:
- based upon the feedback BiomX has received from their cosmetic partner, assuming positive Phase 2 results are obtained in Q2, BX001 will require approximately 18 months of pre-commercial activity (including pre-marketing, manufacturing scale-up, packaging, and related activities) before being commercially launched;
- BiomX has not entered into a definitive commercial agreement for BX001, and has retained all commercial licensing rights worldwide;
- BiomX is evaluating several commercial options including exclusively licensing BX001 to a leading multinational cosmetic company to commercialize BX001 in return for royalty and other payments;
- The first country for the commercial launch of BX001 has not been finalized;
- BiomX has not determined the pricing strategy for BX001 but “the range is usually between $80-$150 for a 50 ml tube”;
- The advantages of BX001 compared to traditional antibiotics include:”Antibiotic resistance – In preclinical studies, BX001 was shown to be active on antibiotic-resistant C. acnes strains.· Penetration of biofilm (a matrix secreted by the bacteria which surrounds them and makes them less accessible to substances such as antibiotics) – In in vitro experiments, phage reduced the number of viable C. acnes bacteria within biofilm by 100,000 fold within 24 hours resulting in undetectable levels after 48 hours. Under the same conditions (using a C. acnes strain sensitive to erythromycin), erythromycin, a common antibiotic, reduced bacterial levels by approximately 100 fold after 48 hours.· BX001 Only kills C. acnes, keeping the rest of the microbiome intact”.
Source: BiomX February 2, 2021 Corporate Presentation
Size of the Global Cosmetic Acne Market
In his February 14th email to me, CBO Assaf Oron indicated that based upon BiomX’s research, the global cosmetic acne market is estimated to be approximately $4 billion, and the global market for antibiotics for acne is estimated at $1 billion.
Mr. Oron also stated that as BX001s mode of action is similar to antibiotics (both are aimed at killing C. acnes bacteria, implicated in the pathophysiology of acne vulgaris), “BX001 could potentially be launched as a cosmetic and compete with the use of existing antibiotics, which have been losing effectiveness due to extensive use in the last decades and gradual emergence of antibiotic resistance.”
2. BX003 (an updated version of BX002)- Phage Cocktail Targeting Gut of IBD and PSC Patients
In his January 11th webcast, Solomon described the idea of being able to successfully treat IBD (irritable bowel disease) as the holy grail of microbiome therapeutics as it represents a $20 billion market. Currently, there is enormous unmet need and microbiome therapeutics is showing some early signs that may provide an answer to that painful and often debilitating disease. IBD is also the reason why Solomon believes both Takeda and J&J were early investors in BiomX.
In discussing the background of the BX003 phage cocktail candidate (an updated version of BX002), Solomon described how researchers in certain laboratories discovered that patients with IBD had much higher levels of the Klebsiella bacterial strain in their gut compared to healthy patients. Furthermore when IBD patients suffered from IBD “flare-ups” their Klebsiella numbers shot up. Conversely, when IBD symptoms went into remission, their Klebsiella numbers plummeted.
In different laboratories, researchers found that patients who suffered from a serious orphan liver disease, primary-sclerosing-cholangitis or PSC, were also showing very high levels of the Klebsiella bacteria strain in their gut (despite PSC being a liver disease) in contrast to healthy patients. Similar to IBD patients, researchers found that when PSC patients suffered flare-ups their Klebsiella bacteria levels had increased dramatically and when the symptoms subsided or went into remission, the levels of Klebsiella plummeted. Researchers also discovered that all PSC patients suffered from IBD.
On further research, it was determined that in PSC patients, the Klebsiella bacteria poked holes in the gut lining (i.e. “leaky gut”) and then caused an inflammation of the liver, often causing so much damage that 1/3 of all PSC patients require a liver transplant. The article itself does not go as far as make a direct link between the existence of the Klebsiella bacteria and the requirement to have a liver transplant.
There is currently no existing approved therapy for PSC so it represents a substantial unmet need and opportunity for BiomX if BX003 is effective.
On November 16, 2020, BiomX presented a poster presentation at the AASLC Conference on using phages as a potential therapy against PSC.
Source: BiomX November 16, 2020 Poster Presentation at AASLC Conference
BiomX has developed a 2nd generation three-phage cocktail which has shown impressive preclinical results in mice in which the phage cocktail resulted in near-complete (99.9%) eradication of bacteria from a single dose, within a few hours. Solomon believes that no antibiotic can do this. See the BiomX slide below for additional details.
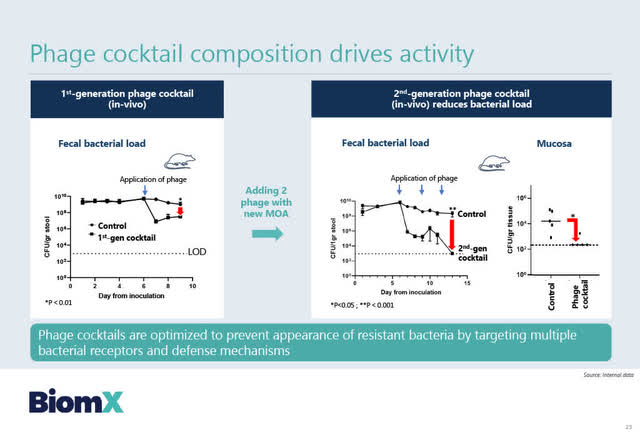
Source: BiomX February 2, 2021 corporate presentation
BX002 was an earlier version of BiomX’s IBD product and has now been replaced by BX003. Prior to November 2020, BiomX had two separate phage product candidates for IBD and PSC.
As set out in BiomX Q3 2020 earnings release and update on November 12, 2020, BiomX announced the consolidation of its IBD and PSC programs and advancing BX003 for both programs as both programs target Klebsiella pneumonia, the pathogen implicated in both diseases.
BX002 Phase 1a Study Results: On February 2, 2021, BiomX issued a press release announcing positive results of its Phase 1a pharmacokinetic study in BX002,
… BX002 is BiomX’s orally administered phage therapy candidate targeting Klebsiella pneumoniae (K. pneumoniae) bacteria in the gut, which have been linked to the pathogenesis of both inflammatory bowel disease (IBD) and primary sclerosing cholangitis (PSC). In the Phase 1a study, BX002 was demonstrated to be safe and well-tolerated, with no serious adverse events and no adverse events leading to discontinuation. In addition, the study met its objective of delivering high concentrations of viable phage to the gastrointestinal tract of approximately 1010 PFU (plaque forming units). This equals approximately 1,000 times more viable phage compared to the bacterial burden of K. pneumoniae in IBD and PSC patients as measured in stool. …”
Source: BiomX February 2, 2021 corporate presentation
Based on the successful Phase 1a results, BiomX has announced that it is moving forward with its Phase 1b/2a study for BX003 which will study the potential of BX003 for reducing K. pneumoniae levels in patients carrying the bacteria. The readout from the Phase 1b/2a study is anticipated in mid-2022.
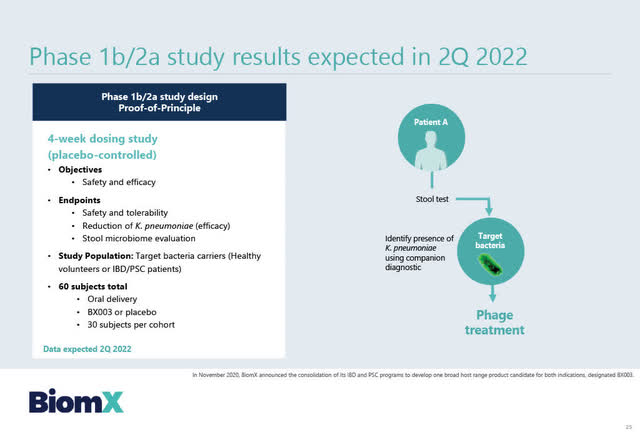
Source: BiomX February 2, 2021 Corporate presentation
Potential Size of IBD and PSC Market in U.S. & Europe
According to the U.S. Centre for Disease Control and Prevention (“CDC”) website Inflammatory Bowel Disease or IBD is a condition “characterized by chronic inflammation of the gastrointestinal tract. The two most common inflammatory bowel diseases are ulcerative colitis and Crohn’s disease. Inflammation affects the entire digestive tract in Crohn’s disease and only the large intestine (also called the colon) in ulcerative colitis. Both illnesses involved an abnormal response to the body’s immune system.”
The CDC reports that in 2015, about 1.3% of more than 3 million Americans suffer from IBD. For more CDC statistics about IBD see CDC’s website page found here.
According to the American Liver Foundation, Primary Sclerosing Cholangitis – Symptoms, Causes, and Treatment,” PSC is a rare progressive liver disorder with no FDA-approved treatment. PSC affects approximately 30,000 patients in the U.S. It is characterized by inflammation and fibrosis of the bile ducts, which often results in the obstruction or interruption of bile flow from the liver, a condition known as cholestasis. Without a liver transplant, patients with PSC have a median survival period after diagnosis of nine to eighteen years. Even following liver transplantation, approximately 40% of patients experienced a recurrence of PSC disease.”
According to BiomX in the slide below, the potential size of the PSC market in the U.S. and Europe is approximately $3.6 billion. Currently, there are no approved therapies for PSC.
Source: BiomX slide attached to February 14, 2021 email to author
Cystic Fibrosis (“CF”)
In its Q3 2020 earnings release on November 12, 2020, BiomX announced a new phage therapy program “targeting chronic respiratory infections caused by Pseudomonas aeruginosa, the main contributor to morbidity and mortality in patients with cystic fibrosis.”
BiomX describes this trial in one of its slides below as a Phase 2 “personalized proof of concept trial” in which personalized phage treatments will be developed by BiomX for each CF patient in the trial. Both the CF Phase 2 program (and the A. Dermatitis program discussed later in this article) are benefiting from BiomX’s BOLT platform. As a result, Phase 2 results of this CF proof of concept clinical study evaluating safety and efficacy in patients are expected in Q4 2021.
As described in BiomX’s Form 10-Q filed November 12, 2020, the BOLT platform allows BiomX to develop a very accelerated program of a personalized phage treatment in only 12 to 18 months from project initiation to Phase 2 clinical proof of concept. BiomX is now testing this program in both Cystic Fibrosis and Atopic dermatitis.
Currently, patients suffering from Cystic Fibrosis suffer from recurring respiratory infections due to Pseudomonas aeruginosa. As set out in the BiomX slide below, over the years these CF patients find that repeated antibiotic dosing leads to multi-drug resistance of the P. aeruginosa bacteria strain, which can lead to death.
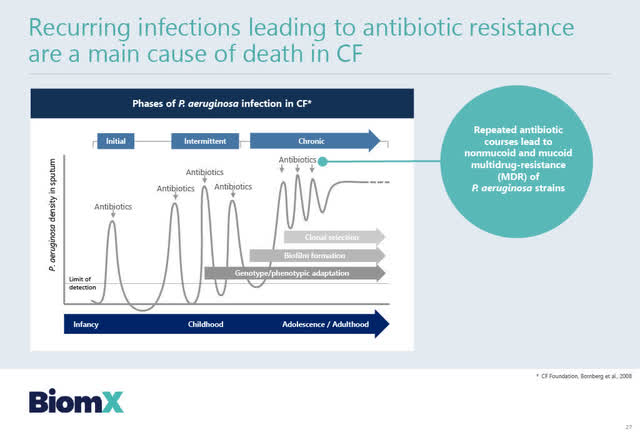
Source: BiomX February 2, 2021 Corporate presentation
Some recent studies and compassionate use cases have found that phage therapies may be effective as a therapy to treat P. aeruginosa pulmonary infections.
In the slide below provided by BiomX to me on February 14, 2021, BiomX summarizes the results of 11 CF patients treated with phage therapies targeting P. aeruginosa with very promising results. One of the studies was conducted was on a group of 8 CF patients at Yale University.
A larger 36 patient Phase 2 CF trial targeting P. aeruginosa at Yale University is scheduled to begin this month. For further details on Yale’s Phase 2 trial see clinicaltrials.gov here.
Source: BiomX Compassionate Use Presentation
Source: BiomX February 2, 2021 corporate presentation
Atopic Dermatitis
BiomX has also initiated a personalized phage-targeted program to treat Atopic Dermatitis, associated with a bacteria called staphylococcal aureus or S. aureus. Atopic Dermatitis is a common severe form of eczema.
Under the BOLT platform, a Phase 2 trial is being planned by BiomX targeting Atopic Dermatitits as set out in the two slides below.
BiomX has indicated that it expects that Phase 2 results of this Phase 2 proof of concept clinical study in Atopic Dermatitis will be available in H1 2022.
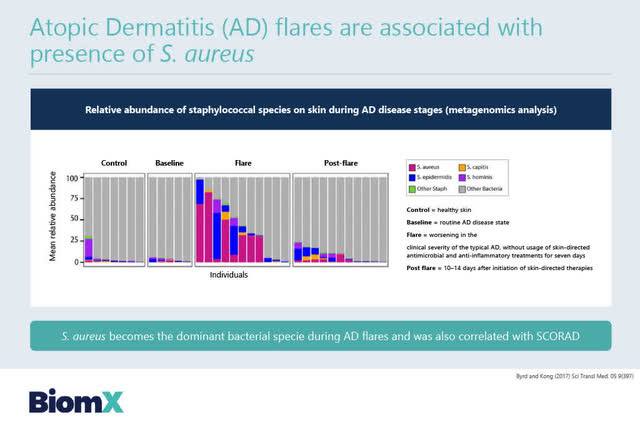
Source: BiomX February 2, 2021 Corporate presentation
Source: BiomX February 2, 2021 Corporate presentation
Colorectal Cancer
BiomX has also begun a preclinical program using engineered phage therapies to treat colorectal cancer tumors. Colorectal tumors are generally unresponsive to immunotherapies.
In his January 11th webcast presentation, Solomon discussed BiomX’s proposed phage therapy program for colorectal cancer, and indicated that one of the keys to making colorectal tumors responsive to immune checkpoint inhibitors is turning the tumor from “cold” to being a “hot tumor”.
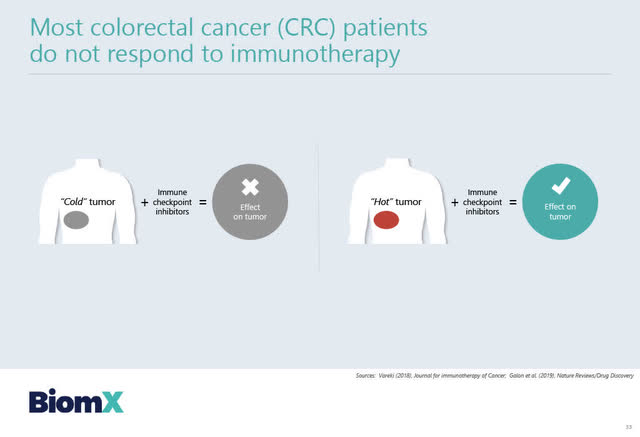
Source: BiomX February 2, 2021 corporate presentation
At ESMO on December 9, 2020, BiomX’s presented a poster publication, “Novel Analysis of Fusobacterium Nucleatum Species in Human Colorectal Cancer and Engineering of Therapeutic Bacteriophage” in which BiomX summarized the role of Fusobacterium nucleatum in colorectal tumors, and that its presence correlates with advanced tumor stage and poor prognosis.”
BiomX is developing engineered phage cocktails in which they are adding multiple synergistic “immune stimulating payloads” as described by BiomX (in its slides below) to target Fusobacterium nucleatum inside the colorectal tumors.

Source: BiomX February 2, 2021 corporate presentation
BiomX believes that theirs is a novel approach; its engineered phages with additional payloads will attack the F. nucleatum directly as a novel target to “uncloak” cold colorectal tumors to “hot”.
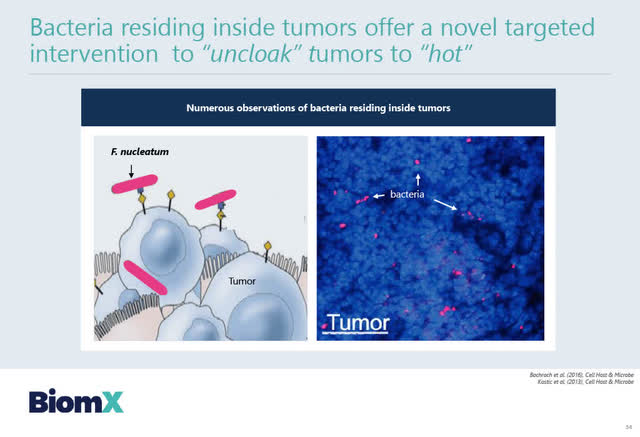
Source: BiomX February 11, 2021 corporate presentation
In preclinical animal studies, BiomX has already been testing these synthetically engineered phages which are delivered by IV directly into the tumor.
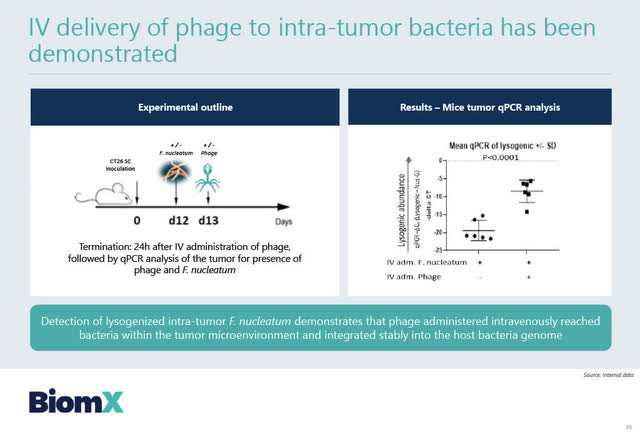
Source: BiomX February 2, 2021 corporate presentation
To date, BiomX’s preclinical in vivo studies have demonstrated that this engineered phage therapy (with multiple anti-tumor payloads) can turn the colorectal tumors from cold to hot tumors. The data is promising, but very early stage.
Ongoing in vivo preclinical studies in this program are continuing with additional results expected by BiomX to be released in H1 2021.
Source: BiomX February 2, 2021 corporate presentation
Upcoming Milestones and Catalysts
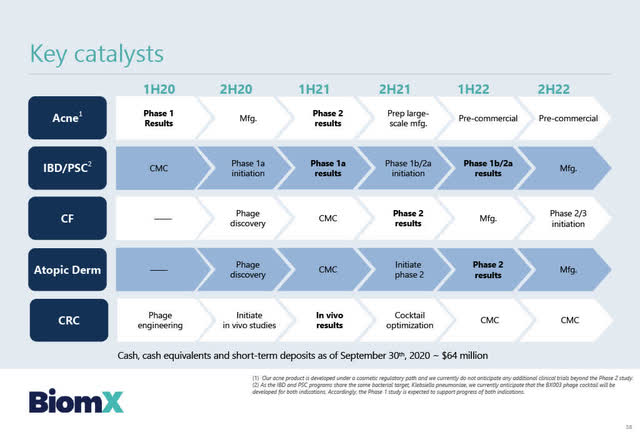
Source: BiomX February 2, 2021 Corporate presentation
BiomX: Upcoming milestones and catalysts include:
- Phase 2 cosmetic clinical study results of BX001 (100 patients with mild-to-moderate acne) to be released in Q2 2021. If the Phase 2 BX001 acne trial is successful, pre-commercial activities for BX001 will commence in H1 2021 lasting approximately 18 months before being launched commercially.
- BiomX entering into a commercial licensing agreement with a large multi-national cosmetic company to launch BX001 commercially for the treatment of mild-to-moderate acne. Based upon email correspondence from BiomX CBO, BiomX believes such a licensing agreement will take about 6 months to negotiate, although there is no assurance that a deal will be finalized within that time frame or at all.
- In vivo pre-clinical test results (in mice) in colorectal cancer testing BiomX’ synthetically engineered phage cocktails with multiple anti-tumor payloads expected to be released in H1 2021.
- Personalized Phase 2 “proof of concept” trial utilizing a BiomX phage cocktail to treat 20 CF patients’ results are expected in Q4 2021.
- Phase 1b/2a clinical trials testing BX003 to treat IBD/ PSC to begin in H2 2021 with anticipated results in H1 2022.
- Phase 2 trial for the treatment of A. Dermatitis to begin in H2 2021 with results expected in H1 2022.
- Assuming Phase 2 “proof of concept” trial results in CF are positive, Phase 2/3 trial for the treatment of CF to begin in H2 2022.
Analysts Share Price Targets (currently trades in the $7.40 range)
According to a recent Bloomberg report,
1. Cantor Fitzgerald – $19.00 target
2. Chardan Capital – $28.00 target
Largest Institutional Shareholders (according to recent Bloomberg report)
1. Takeda Pharmaceuticals Co. Ltd. – 2,470,935 shares – 10.78%
2. Orbimed Advisors LLC – 2,313,489 shares – 10.10%
3. Johnson & Johnson – 2,133,402 shares – 9.31%
4. MMCap International Inc.* – 684,451 shares – 2.99%
*MMCap filed a Form 13G last week with the SEC effective December 31, 2020 in which it disclosed ownership of an additional 1,431,188 warrants exercisable for an additional 715,594 common shares in BiomX.
Largest Insider Shareholders (according to recent Bloomberg report)
1. Jonas Grossman (Director of BiomX, President of Chardan Capital) – 335,217 shares – 1.45%
2. Kerry Propper (Co-Founder and Chairman of Chardan Capital) – 260,771 shares – 1.14%
3. Gbolahan Amusa (Director of BiomX, Head of Healthcare Research at Chardan Capital) – 237,871 shares – 1.04%
Other Phage Based Biotech Companies
As far as I am aware there are currently no phage therapies that have been approved by the FDA or other major regulatory agencies in other jurisdictions.
In reviewing other phage-based biotech companies, almost all of them are relatively early stage and nearly all are still private. The “phage based” biotech companies include Adaptive Phage Therapeutics (private), Armata Pharmaceuticals, Inc. (NYSE: ARMP), Cytophage Technologies (private), Eligo Bioscience (private), Enbiotix, Inc. (private), Felix Biotechnology (private), Intralytix (private), PhagePro, Inc. (private), PhagoMed Biopharma (private), Pherecydes Pharma (private) and TechnoPhage (private).
Investment Thesis Conclusion
BiomX Inc. (PHGE) is using its proprietary synthetic biology XMarker and BOLT platforms to develop and clinically test phage cocktails (both naturally occurring or engineered or in some combination thereof) as a novel mode of action to potentially treat various chronic and other diseases.
BiomX currently has 6 pipeline targets including acne, IBD, PCF, CF, A. Dermatitis, and colorectal cancer. Each of the target areas represents an enormous unmet need and represents a number of potential multi-billion-dollar markets.
Early pre-clinical and clinical results have been promising and BiomX has a number of upcoming catalysts and milestones in 2021 and 2022.
The BOLT platform permits for an accelerated personalized phage development program which is a novel approach, and which has enormous treatment and commercial value if successful. As a result, BiomX pipeline programs for CF and A. Dermatitis are expected to have Phase 2 trial results in late 2021 and H1 2022 respectively.
BX001 has the potential to be commercially launched in early 2023, likely under a licensing agreement with a large multinational cosmetics company (to be determined) which will likely generate royalty and other revenues that are potentially substantial given the sizes of the acne market and the premium price positioning BX001 might obtain assuming successful Phase 2 clinical trial results are generated.
With a promising pipeline, an experienced management team and board of directors, shareholders that include Orbimed, Takeda, and Johnson & Johnson as well as collaboration agreements with Janssen, Boehringer Ingelheim, and others, BiomX has enormous commercial value and potential.
In the author’s opinion, BiomX is substantially undervalued at the current share price of approximately $7.40 with a market cap of approximately $170 million and nearly $50 million in cash or equivalents as of September 30, 2020.
I expect to see one or more equity or other financings in 2021 given the filing of both its December 4, 2020, $50 million ATM with Jefferies and its $150 million shelf prospectus filed in December 2020. Management has already guided in its Form 10-Q that it has sufficient cash to last until mid-2022 so equity financing in 2021 should be expected.
If BiomX’s clinical and pre-clinical programs demonstrate success during the next 12 to 24 months, BiomX shares could be worth a multiple of its current price despite some dilution along the way.
Investment Risks
An investment in BiomX has numerous risks. It is an early-stage biotech company without any current or predictable revenues. Investment risks include, but are not limited to, the possibility of clinical trial failures, the inability to recruit patients in a timely fashion, the inability to negotiate a reasonable licensing agreement for BX001, competition, the inability to obtain or maintain certain patents, IP infringements and challenges, loss of key personnel, share dilution, and general market risks.
Management has provided guidance that BiomX has sufficient cash until mid-2022 so investors should expect one or more additional financings during the next 12 months and in light of its $50 million ATM with Jefferies and $150 million shelf prospectus filed in December 2020 with the SEC.
Phage cocktails are a relatively new area of therapeutic development that is still unproven.
For further details of the potential risks involved, see the risk factors set out in the company’s most recent regulatory filings including the Company’s Form 10-Q filed in November 2020 with the SEC and available on BiomX’s website, and the EDGAR website.
BiomX should be considered a high-risk investment in which an investor could lose all or part of his or her investment. Before investing in BiomX, you should do your own due diligence and obtain professional advice to determine whether it is an appropriate investment for you and the sizing of such an investment.
